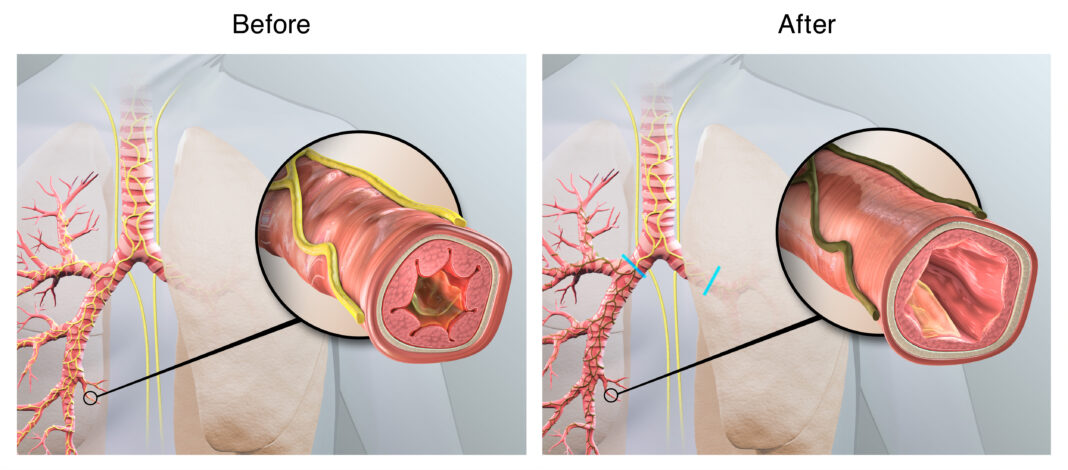
LOUISVILLE, Ky. – Physicians from the University of Louisville are the first in Kentucky to perform an investigational procedure designed to help patients with chronic obstructive pulmonary disease, or COPD. The procedure, known as targeted lung denervation (TLD), may provide an additional treatment option for patients suffering from frequent COPD flare-ups, or lung attacks.
COPD causes restricted airflow from the lungs, resulting in breathing difficulty, cough, mucus production and wheezing. TLD involves the removal of tissue in the lungs that may be making symptoms worse. Performed via bronchoscopy, the procedure is designed to disrupt pulmonary nerve reflexes, which may have the potential to reduce COPD exacerbations.
AIRFLOW-3, a phase 3 clinical trial sponsored by Nuvaira, is enrolling patients with moderate-to-severe COPD, high symptom burden and a history of COPD exacerbations, including increased coughing, wheezing and breathing distress, to evaluate the safety and effectiveness of TLD.
Umair A. Gauhar, M.D., associate professor at the UofL School of Medicine, led a medical team in the Division of Pulmonology that successfully treated a COPD patient with TLD therapy in one-hour-long outpatient procedure at UofL Health – UofL Hospital. The patient returned home the same day.
“Many of our COPD patients experience exacerbations which cannot be controlled with their inhaler medications,” Gauhar said. “As a pulmonary community, we need to embrace the development of interventional therapies which may stabilize COPD patients and help reduce their risk of exacerbation. Based on the existing evidence, we are enthusiastic about the potential of TLD to meet a true unmet medical need and are proud to have treated our first patient in the AIRFLOW-3 clinical trial.”
Debbie Sowers, a 58-year-old resident of Madison, Ind., said she participated in the trial at UofL to help develop better treatments for COPD.
“I participated to help everybody that has COPD and to try to better my breathing,” Sowers said. “There need to be more studies on COPD because it is a really bad disease. It stops your life.”
UofL is the only center in Kentucky enrolling COPD patients for AIRFLOW-3 and is one of up to 25 centers across the United States enrolling patients in the trial.
Nuvaira is headquartered in Minneapolis, Minn. The company’s proprietary Nuvaira® Lung Denervation System addresses airway hyper-responsiveness, a pathophysiologic underpinning of both COPD and asthma, with TLD therapy. The Nuvaira Lung Denervation System is an investigational device in the United States and has CE mark approval in the European Economic Area (EEA). Nuvaira has completed and published four clinical studies, including AIRFLOW-2, a randomized, sham-controlled, double-blind, multicenter clinical trial.
#WeAreUofL























