LOUISVILLE, Ky. – Jun Yan, M.D., Ph.D., professor of surgery and microbiology and immunology at the University of Louisville, led a team of researchers in the discovery of an important biomarker that predicts a crisis in COVID-19 patients that could lead to death.
Yan, an immunologist and researcher with the UofL Health – James Graham Brown Cancer Center, along with UofL Professor of Anesthesiology Jiapeng Huang, M.D., Ph.D., and M.D./Ph.D. student Samantha Morrissey, Ph.D., conducted a patient study to better understand the most severe cases of COVID-19.
Approximately 20 percent of COVID-19 patients experience severe disease, including pneumonia and acute respiratory distress syndrome (ARDS). In some of these patients, the rapid influx of immune cells to the lungs to fight the infection leads to complications in the lung and blood coagulation disorders, resulting in heart attack, pulmonary embolism, stroke or deep vein thrombosis.
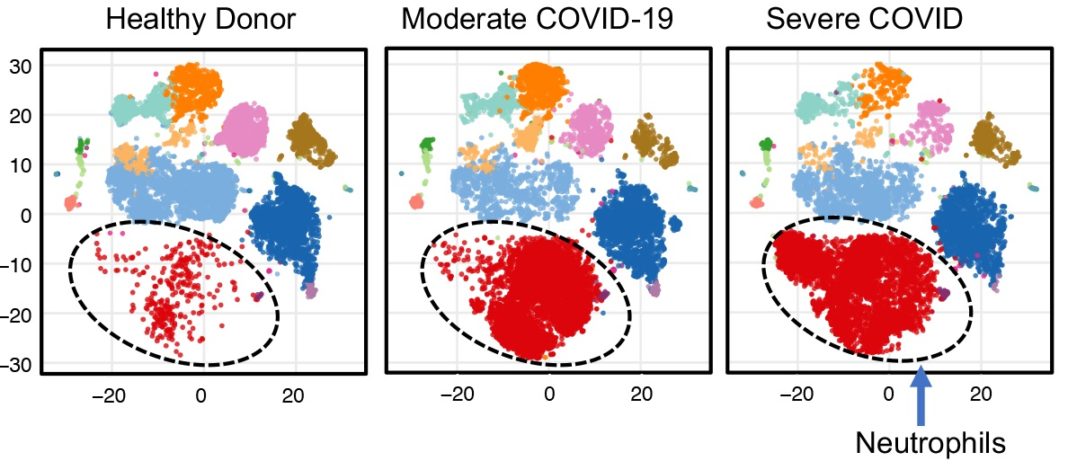
To better understand these serious complications, Yan’s team evaluated levels of various immune cells in blood samples of COVID-19 patients and compared those levels with those of healthy individuals. They discovered that one type of immune cells, low-density inflammatory neutrophils, became highly elevated in some patients whose condition became very severe. This elevation signaled a point of clinical crisis and increased likelihood of death within a few days.
Neutrophils are one type of immune cells that serve as the first line of defense in the body. When an individual acquires an infection, neutrophils rush to the site to clear the pathogen causing the infection. However, if their presence is persistent or there is a very high volume of cells at the site of infection, in this case the lungs, they can cause unwanted adverse effects. In the case of patients with severe COVID-19, a blood clotting disorder known as coagulopathy occurred, that can manifest as pulmonary embolism, heart attack or stroke.
The study, published online as a preprint, details shifting levels of these neutrophils and other immune cells through repeated blood samples from study participants, correlated with improvement or worsening of the patients’ condition. If clinicians could detect a rise in these cells, they may be able to provide therapy to prevent the potential life-threatening conditions associated with them.
“Based on this study, we believe that the low-density inflammatory band neutrophil population contributes to COVID-19-associated coagulopathy (CAC) and could be used as a clinical marker to monitor disease status and progression,” Yan said. “Identifying patients who are trending toward a cellular crisis and then implementing early, appropriate treatment could improve mortality rates for severe COVID-19 patients.”
To provide additional clinical options for physicians in addressing these crises, Yan is now working with other researchers at UofL to test potential therapies.
#WeAreUofL
























